Community Tip - Did you get called away in the middle of writing a post? Don't worry you can find your unfinished post later in the Drafts section of your profile page. X
- Subscribe to RSS Feed
- Mark Topic as New
- Mark Topic as Read
- Float this Topic for Current User
- Bookmark
- Subscribe
- Mute
- Printer Friendly Page
Balancing the pH Balance and Composition of Two Chemical Solutions
- Mark as New
- Bookmark
- Subscribe
- Mute
- Subscribe to RSS Feed
- Permalink
- Notify Moderator
Balancing the pH Balance and Composition of Two Chemical Solutions
By D.M. Griffin, Jr.
Using Repetitive Solve Blocks in Chemistry Problems
- Determines at what pH the composition NaOH added to water saturated with CO₂ at a partial pressure of 10⁻3.5 atm will be the same as a solution made only of Na₂CO₃ and water
- Applicable in chemistry, and those learning PTC Mathcad
- Performs mass balance, charge balance, proton condition, solve blocks, "trace", graphing, error analysis
Determines at what pH the composition (Sodium Hydroxide, NaOH, added to water saturated with CO₂ at a partial pressure of 10⁻3.5 atm) will be the same as a solution made only of Na₂CO₃ and water. This worksheet shows you how you can use PTC Mathcad to solve complex chemistry problems. You can use repetitive solve blocks to look at the spread in the values on the plot and many orders of magnitude, typical in chemistry problems. Instead of doing this problem by hand you can easily input, edit, and arrange different sets of the data in PTC Mathcad.
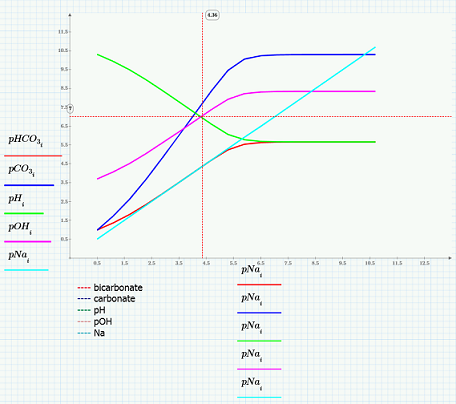
This worksheet first lists the equations necessary including the mass balance on Na and the charge balance. Next the worksheet identifies the proton condition and lists the terms of the given and unknown elements. Next, the worksheet shows how you can define Na as the solution of the solve block and identify the independent variable. The bicarbonate, carbonate, pH, pOH, and Na are all plotted and observations can be made.
Using "trace" you can determine how much more concentrated NaOH you can add before the solution pH begins to change and before it reaches 7.0. All elements, data, formulas, notation, and graphs are included to aid you in your own chemistry problems.
Download and explore this worksheet yourself! You can download a free lifetime copy of PTC Mathcad Express and get 30 days of full functionality.


