As Valery already pointed out that formula in your Appendix is not quite clean concerning units. The example given there is calculated dimensionless, as Valery has done.
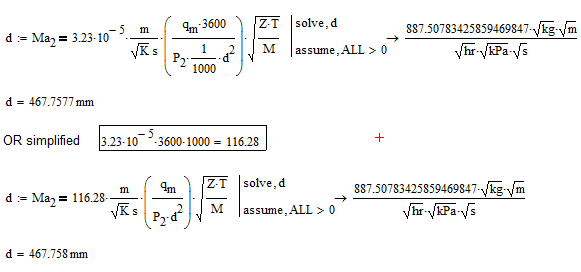
I would correct the fact, that Ma2 has no unit by assigning the constant 3.23*10^(-5) the appropriate unit. I'm not sure about units of Z and esp. M !??
The reason you are not getting the answer you expect is, that qm and P2 are not using basic SI units. You may compensate by multiplying qm by 3600 (kg/s --> kg/hr) and dividing P2 through 1000 (Pa --> kPa). That way you have at a formula with a constant of 116.28 you may use with data of any units (as long as your worksheets units are set to SI units in preferences).
To get only the positive solution you would normally use the modifier "assume, d>0", but the symbolic engine does not know anything about units at all. Thats the reason you see kPa, m etc. in the symbolic result - the symbolic engine sees them as unknown variables. Because of that I had to use "assume, ALL>0" to make it work.
Hope it helps.
BTW, is there a specific reason you are appending the literal subscript point to the names of all variables which do not use subscripts ("Z." instead of "Z", etc.)?


